Heat, fuel, oxygen
The science of fire
Fires need three things at once: heat, fuel, and oxygen. Friction kits add heat by rubbing wood; dry fuel catches when it reaches its ignition temperature; oxygen from the air feeds combustion. The ideas are real science—explained here in plain language—and every experiment belongs outdoors with an adult.

The Fire triangle
Picture a triangle with three corners. Each corner is required for a sustained flame: heat to get the reaction started, fuel to burn, and oxygen (usually from the air) to keep Combustion going. Remove any one corner—cool the fuel, cut off air, or run out of burnable material—and the fire cannot keep going the same way.
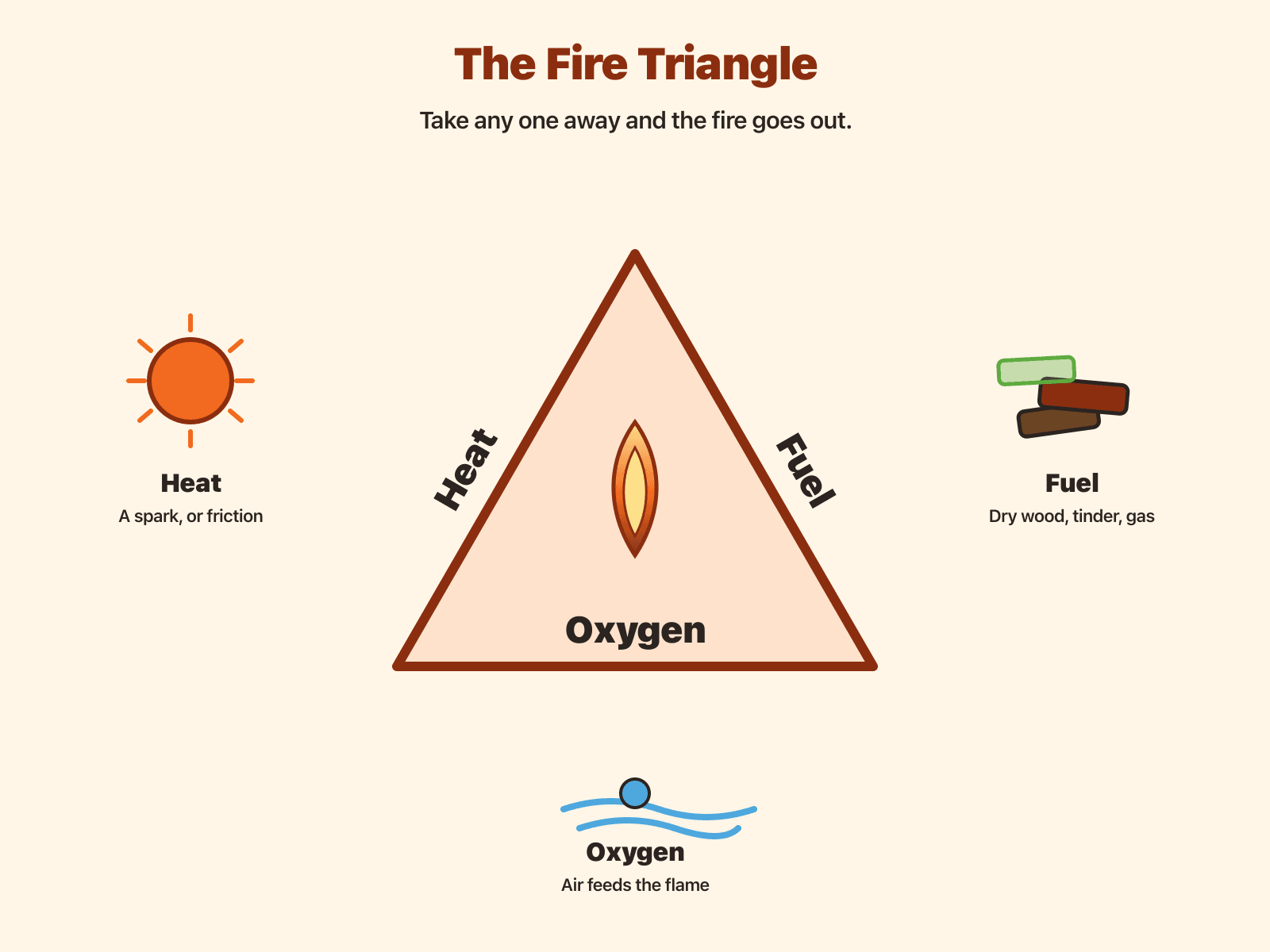
Heat
Heat wakes fuel molecules up until they can react with oxygen. Rubbing wood converts motion energy into thermal energy; matches and lighters do the same job with chemistry instead of friction.
Fuel
Fuel is anything that can oxidize fast enough to glow or flame—here, mostly dry plant material sized from powdery tinder up to larger sticks.
Oxygen
Air is roughly one-fifth Oxygen . A gentle breath can add oxygen to a coal, but timing and aim matter, which is why adults coach the motion.
Friction turns motion into heat

When two surfaces rub, microscopic bumps catch, break, and re-form. That work becomes heat in the contact zone. A bow drill and a Hand drill both concentrate friction where the spindle meets the fireboard so wood dust can climb toward its Ignition temperature . The feeling of warm hands after a brisk rub is the same physics on a gentler scale.
Friction does not guarantee fire: the wood must be dry, the geometry steady, and the pace long enough for heat to build faster than it leaks into the air. That is why patience is a science skill, not only a character trait.
Ignition temperature and the first glow
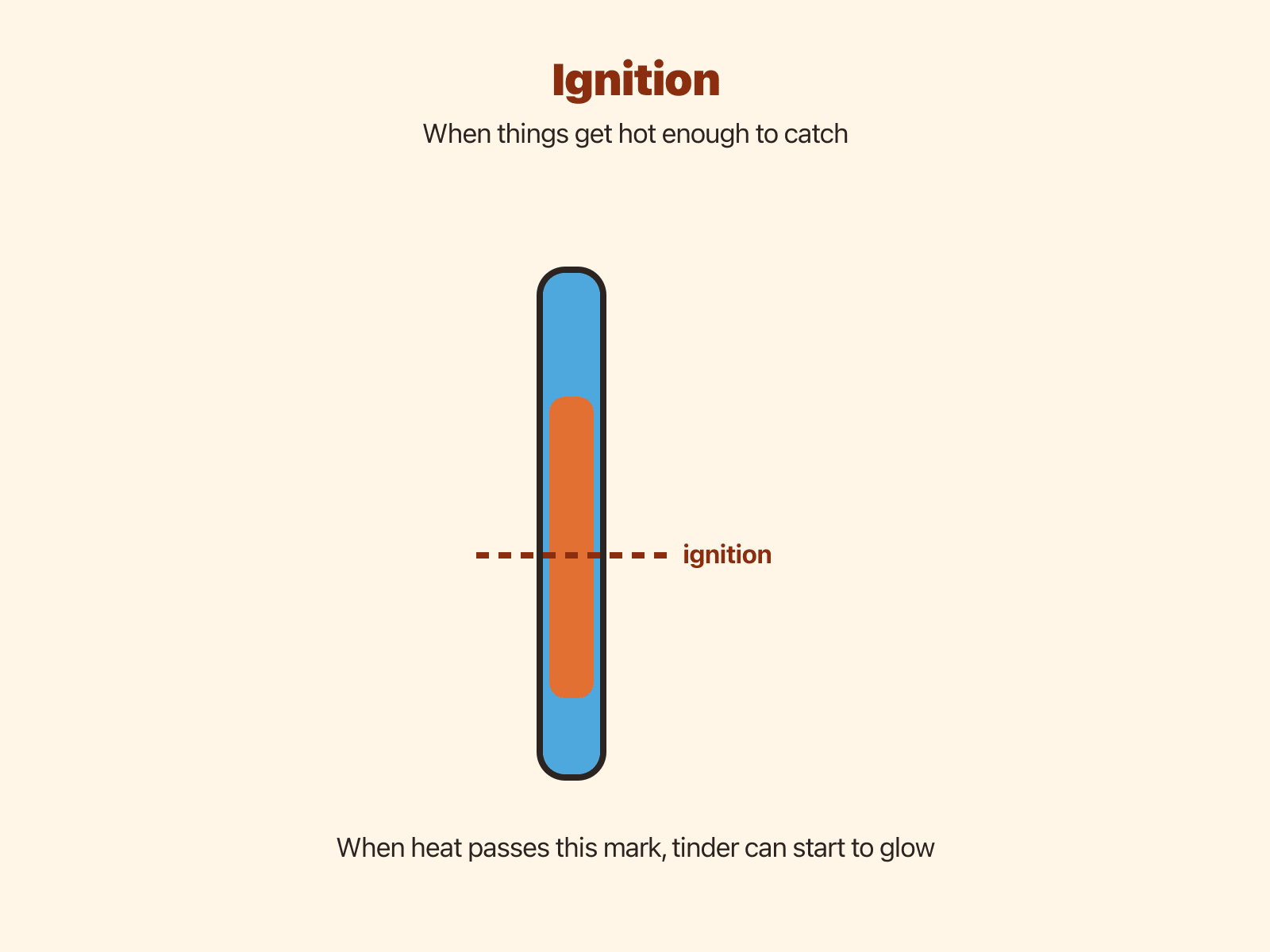
Every fuel has an Ignition temperature —the approximate temperature where enough vapor or char reacts with oxygen that a glow can sustain itself. Exact numbers depend on moisture, chemistry, and airflow, so outdoor learners focus on dry material, sharp notches, and steady technique rather than memorizing digits.
Before a flame appears, a hot Ember often shows up: a tiny bed of char that radiates heat without tall flames. Treating that stage with respect prevents accidental spreads.
Combustion and Oxidation

Combustion is the rapid chemical reaction that releases heat and light when a fuel combines with oxygen—what people mean by “burning” in everyday language. Oxidation is the broader chemists’ term for losing electrons to an oxidizer (here, oxygen gas). Rust is slow oxidation; a campfire is fast oxidation that human eyes call fire.
Once tinder is hot enough, Oxidation speeds up, releasing more heat, which in turn drives neighboring fuel toward its Ignition temperature . That feedback loop is why small mistakes near dry grass escalate quickly—and why adult supervision and cleared ground matter.
Tinder , Kindling , and larger fuel
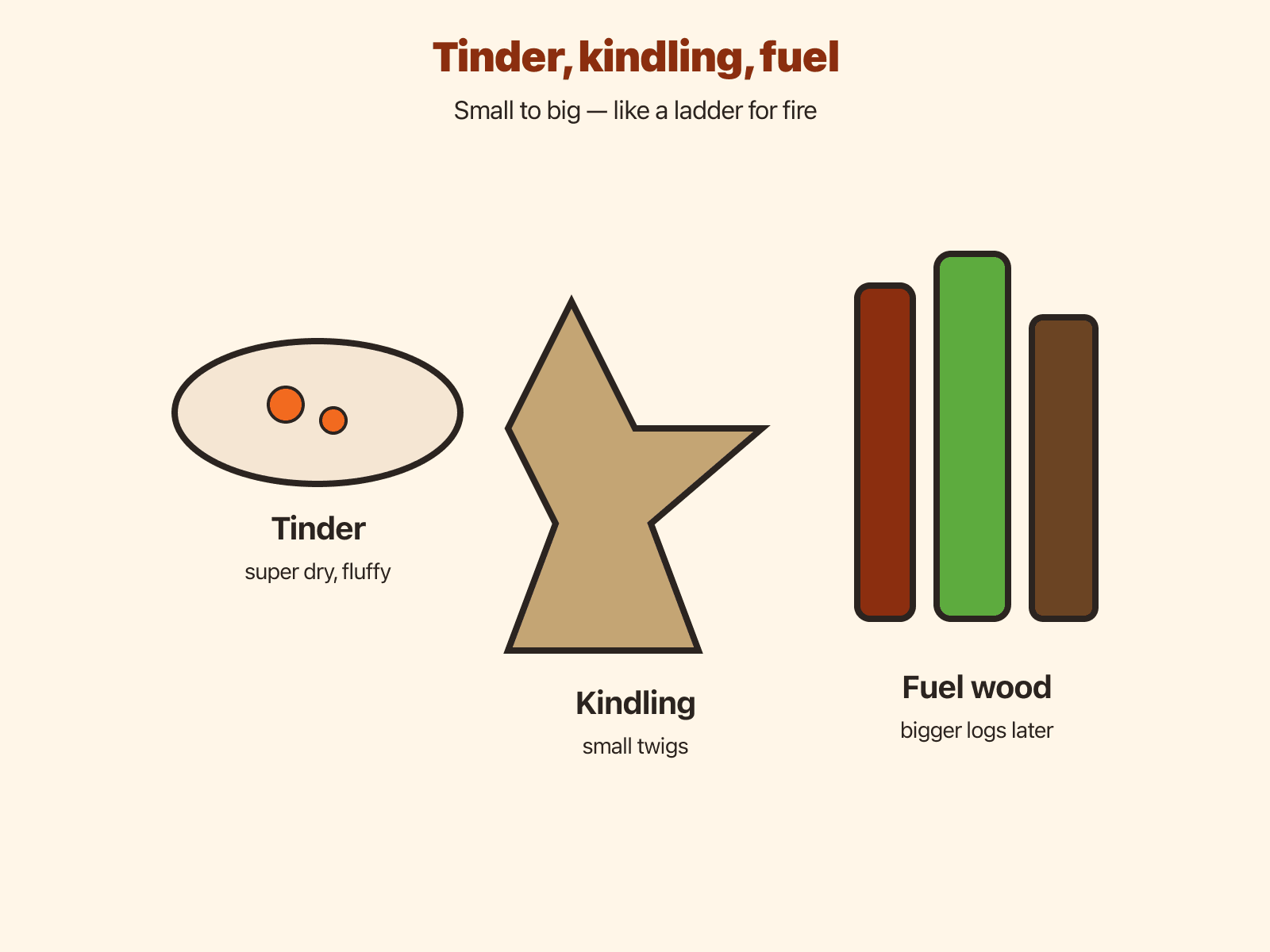
Tinder is the finest, driest layer—cottony fibers, shaved bark, or other fluff that lights from a coal. Kindling bridges the gap between fluff and thumb-thick sticks. Larger logs belong to established fires managed by adults with mature coals. Skipping sizes usually wastes heat and teaches frustration more than physics.
Learn more
These links open in a new browser tab so you can keep reading here.
- What is fire? (opens in a new tab) — NASA Climate Kids
- Combustion (opens in a new tab) — Britannica
- Oxidation–reduction reaction (opens in a new tab) — Britannica
- Heat and temperature (opens in a new tab) — Khan Academy